3D Bioprinting Market to Reach USD 8.45 Billion by 2032 at 20.07% CAGR: Maximize Market Research
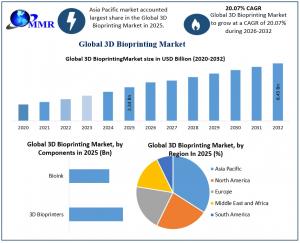
The 3D Bioprinting Market was valued at USD 2.34 billion in 2025 and is projected to reach USD 8.45 billion by 2032, growing at a CAGR of 20.07%.
Will 3D bioprinting end animal testing? Maximize Market Research map the journey through the FDA Modernization Act 2.0.”
ROCKVILLE , MD, UNITED STATES, March 27, 2026 /EINPresswire.com/ -- The 3D Bioprinting Market landscape has transitioned from a theoretical research niche into an industrial-scale clinical reality. While the market entered 2025 with a USD 2.34 Billion valuation, the current momentum is driven by the Moonshot shift in organ-on-demand manufacturing, exemplified by the 2026 ImPLANT project milestones. As regulatory barriers dissolve under the FDA’s Modernization Act 2.0, the pivot toward animal-free drug toxicity screening and immunocompetent liver tissue fabrication is propelling the industry toward a USD 8.45 Billion peak by 2032.— Maximize Market Research
Get Full PDF Sample Copy of Report: (Including Full TOC, List of Tables & Figures, Chart) @ https://www.maximizemarketresearch.com/request-sample/3271/
This 20.07% CAGR is not merely a reflection of sales, but of a fundamental breakthrough in suspension printing and vascular network maturity. The integration of AI-driven lithographic crystallinity regulation (CRAFT) has solved the longstanding cell-viability bottleneck, allowing for the rapid assembly of multi-layered biological structures. For healthcare providers, the next decade marks the end of static models, as Maximize Market Research identifies the surge in patient-specific cardiac patches and vascularized liver organoids as the primary engines of the USD 8.45 Billion revenue curve.
The AI-Bio-Ink Convergence: Solving the Vascularization Paradox
The primary hurdle in 3D bioprinting has shifted from structural formation to biological viability. The industry is moving beyond static scaffolding toward dynamic bio-inks that mimic the human extracellular matrix. The current breakthrough lies in AI-driven lithographic optimization, where machine learning algorithms predict cellular oxygen consumption to design intricate capillary networks in real-time. This prevents the necrotic core issue where internal cells die due to lack of nutrients.
Illustrating this shift, a March 2026 collaboration between CollPlant Biotechnologies and the Mayo Clinic successfully utilized rhCollagen-based bio-inks to create the first fully humanized 3D skin models that outperformed traditional Matrigel in structural consistency. By integrating AI to regulate the viscoelasticity of these non-animal-derived inks, researchers have moved from printing simple patches to complex, multi-layered tissues. This transition from layer-by-layer deposition to volumetric bioprinting is reducing fabrication times by 80%, ensuring that the USD 8.45 Billion market is anchored in scalable, clinically-ready technology rather than laboratory prototypes.
The Preclinical Transformation: 3D Models Replacing Animal Testing
The most significant structural shift is the rapid abandonment of traditional 2D cultures and animal models in favor of human-relevant 3D bioprinted tissues. This transition is legally bolstered by the FDA’s guidance, which formally outlines the validation of non-animal models for drug safety. By utilizing bioprinted organoids, pharmaceutical companies are identifying human-specific toxicities that were previously undetectable in mice or primates.
A definitive example is the adoption of ‘Liver-on-a-Chip’ systems by major biopharma players, who are now integrating these 3D models into their Investigational New Drug (IND) applications to predict cytokine release syndromes. These human-centric platforms offer 87% higher sensitivity in drug-induced liver injury (DILI) detection compared to legacy methods. This regulatory backed evolution ensures the market's trajectory is driven by high margin industrial applications in drug discovery rather than just academic research.
Get Full PDF Sample Copy of Report: (Including Full TOC, List of Tables & Figures, Chart) @ https://www.maximizemarketresearch.com/request-sample/3271/
Regional Divergence: The Shift Toward Regulatory Sovereignty
3D bioprinting is being redrawn by regional Regenerative Corridors that prioritize clinical speed over traditional R&D. While North America maintains a 38% market share due to the ARPA-H PRINT initiative, the Asia-Pacific region is the growth leader, exhibiting an 18.35% CAGR. This surge is catalyzed by Japan’s aggressive conditional approval pathways, which allow bioprinted tissues to reach patients faster than in Western markets.
A pivotal instance is Japan’s ‘ImPLANT’ milestones, where localized bioprinting hubs in Osaka are now integrated directly into hospital workflows for patient-specific skin and cartilage grafts. Simultaneously, India is emerging as a Bio-Ink Powerhouse, with a 20% annual expansion in its domestic biomaterial sector. Meanwhile, the European market is pivoting toward the Horizon Europe 2026 Work Programme, which has allocated specialized funding for Vascularized Organ-on-a-Chip standards. This regional fragmentation ensures that market dominance is no longer about global volume, but about which geography successfully integrates bioprinting into active surgical practice.
Technical Architecture: Inkjet Precision vs. Magnetic Levitation
The 3D bioprinting hierarchy is defined by a pivot from structural scaffolding toward biological functionality. Inkjet-based bioprinting currently holds a 44.4% market share, favored for its high-speed, non-contact deposition that preserves cell viability during the fabrication of complex tissue interfaces. However, the most disruptive growth is seen in Magnetic Levitation, which eliminates over 80% of common printing errors by using magnetic fields to assemble cells without solid supports.
A transformative real-world application is the use of MIT’s ‘MagMix’ system, which integrates magnetic propellers within bioprinter syringes to prevent cell sedimentation. This technology ensures uniform cell distribution during long-duration prints, directly addressing the necrotic core issue where internal cells die due to nutrient starvation. By combining these precision-drop technologies with label-free magnetic bioassembly, manufacturers are transitioning from laboratory prototypes to viable, vascularized organoids. This technical evolution ensures that the market's high CAGR is anchored in repeatable, clinical-grade production standards rather than simple 3D modeling.
3D Bioprinting Market Segmentation: The Convergence of Hardware and Biomaterials
The worldwide bioprinting ecosystem is bifurcated by technical modality and material complexity. By Technology, the market is currently dominated by Inkjet-based bioprinting, which holds a 44.4% revenue share due to its high-resolution precision and cost-effectiveness. However, the fastest-growing niche is Magnetic Levitation, which is projected to expand at a 13.7% CAGR as it eliminates 80% of common printing errors by assembling cells without solid scaffolds.
Based on Component, 3D Bioprinters remain the largest segment at 62%, but Bio-inks are the primary growth engine, surging as a repetitive-purchase consumable. By Material, Living Cells lead with a 38.7% share, though Hydrogels are expanding rapidly due to their ability to mimic the human extracellular matrix. Geographically, while North America holds a 36.7% share, the Asia-Pacific region is velocity leader, driven by clinical reforms in Japan and India that are transitioning bioprinting from research labs into active surgical practice.
Expert Review: The Commercialization of Functional Bio-Systems
The shift in 3D bioprinting is defined by the transition from morphological mimicry to physiological functionality. As we move toward the USD 8.45 Billion milestone, the primary catalyst is the integration of 4D bioprinting where stimuli-responsive materials allow tissues to mature post-print. This evolution, coupled with the FDA’s acceptance of bioprinted liver models for IND applications, has moved the technology from academic labs into high throughput pharmaceutical pipelines. The 20.07% CAGR is now a reflection of realized clinical utility and the industrial scale reduction of animal testing dependencies.
3D Bioprinting Market key players
EnvisionTEC Inc.
Organovo Holdings, Inc.
Vivax Bio LLC
BICO Group AB
Advanced Solutions Life Sciences LLC
Allevi Inc.
Revotek Co. Ltd.
Precise Bio
3D Systems Corporation
Stratasys Ltd.
Aspect Biosystems Ltd.
Cyfuse Biomedical K.K.
RegenHU
Poietis
CollPlant Biotechnologies Ltd.
Get access to the full description of the report @ https://www.maximizemarketresearch.com/market-report/global-3d-bioprinting-market/3271/
FAQs:
What drives the 20.07% CAGR?
Ans: Growth is anchored in the Clinical Gap. As organ failure rates rise, demand for vascularized functional tissues outpaces supply. This structural deficit, alongside AI-optimized bio-inks, ensures sustained market valuation.
How does the FDA Modernization Act 2.0 impact the market?
Ans: This milestone allows New Approach Methodologies (NAMs) to replace animal testing. Consequently, Big Pharma is redirecting billions into bioprinted human-on-a-chip systems for high-fidelity toxicity screening and faster regulatory approvals.
Why is Inkjet technology dominant?
Ans: Holding a 44.4% share, Inkjet systems offer unmatched precision-drop resolution. Their ability to maintain cell viability during high-throughput biofabrication makes them the industrial benchmark for complex tissue interfaces.
What is the significance of Magnetic Levitation?
Ans: As the fastest-growing niche, Magnetic Levitation uses scaffold-free assembly to eliminate 80% of printing errors, facilitating the rapid maturation of dense, vascularized organoids.
Related Reports
Tissue Engineering Market: https://www.maximizemarketresearch.com/market-report/global-tissue-engineering-market/73928/
Tissue Expanders Market: https://www.maximizemarketresearch.com/market-report/tissue-expanders-market/185641/
Tissue Diagnostic Market: https://www.maximizemarketresearch.com/market-report/global-tissue-diagnostics-market/113670/
Tissue Sealants Market: https://www.maximizemarketresearch.com/market-report/global-tissue-sealants-market/77797/
Regenerative Medicine Market: https://www.maximizemarketresearch.com/market-report/global-regenerative-medicine-market/35229/
Top Reports:
Cowboy Boots Market
Rubber Tire Market
About Maximize Market Research
Maximize Market Research is a global business intelligence firm empowering Fortune 500 companies across 45 countries. We provide high-impact, data-driven strategic intelligence to navigate industrial shifts and secure market dominance.
Domain Focus: Life Science
Our research deciphers the global transition toward vascularized functional tissues and AI-driven bio-ink synergy. We analyze the intersection of New Approach Methodologies (NAMs) and organ-on-a-chip architectures, evaluating high-value shifts in the 3D Bioprinting Market.
Lumawant Godage
MAXIMIZE MARKET RESEARCH PVT. LTD.
+91 96073 65656
email us here
Visit us on social media:
LinkedIn
Instagram
Facebook
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.